Birth control that works with your body
Join a paid research study to try hormone-free, non-invasive, investigational birth control from Flo, the #1 women’s health app.
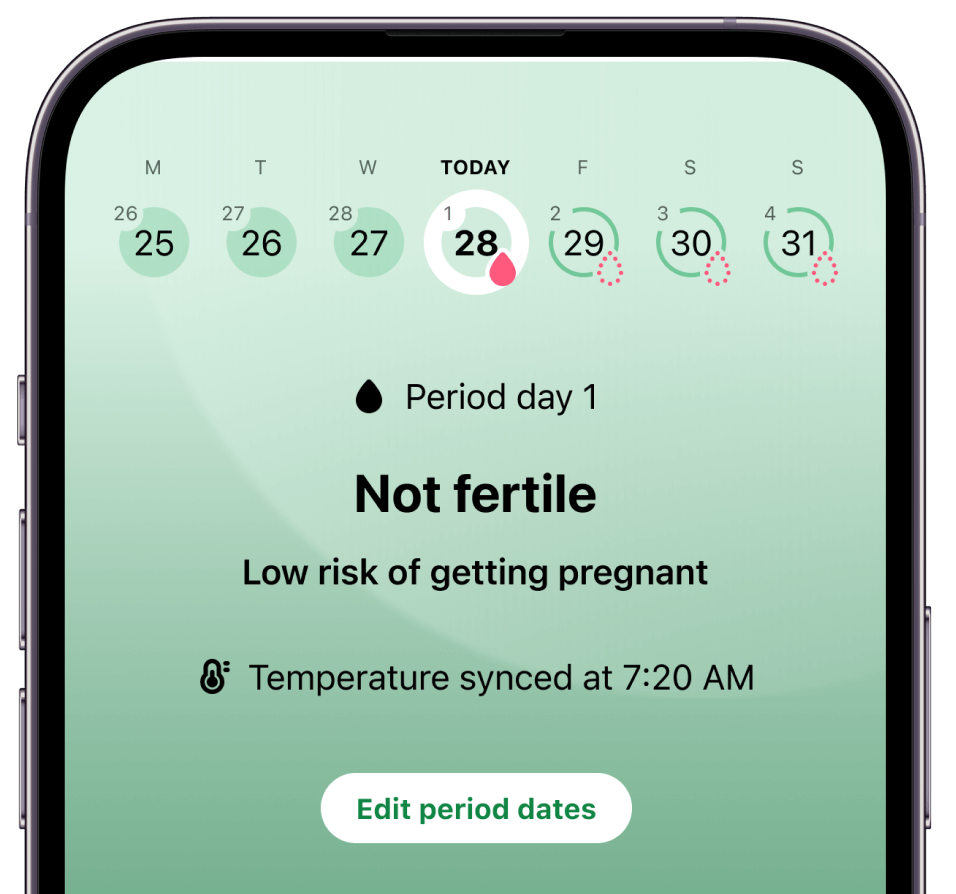
Why join Flo’s birth control study
Pregnancy risk predictions delivered daily
Powered by doctors, backed by science
Enjoy Flo Premium for free (valued at $120)
Digital Contraception is typically 93% effective
According to the FDA, digital contraception can be as effective as the pill for preventing pregnancy with typical use*.
Our study tests the effectiveness of Flo’s new investigational digital contraceptive, which uses Apple Watch temperature data and menstrual cycle data to predict pregnancy risk.

Birth control, on your terms

No hormones. No medication.
Prevent pregnancy without the pills, IUDs, injections, implants, or patches. Just wear your Apple Watch to bed, log your periods and voila! Flo shows you what days to use protection.

Get 2 years of Flo Premium for FREE
Make the most of Flo Premium with personalized insights and in-depth reports, valued at $120. Answer a monthly 5-minute survey to earn up to $90 on top of that.
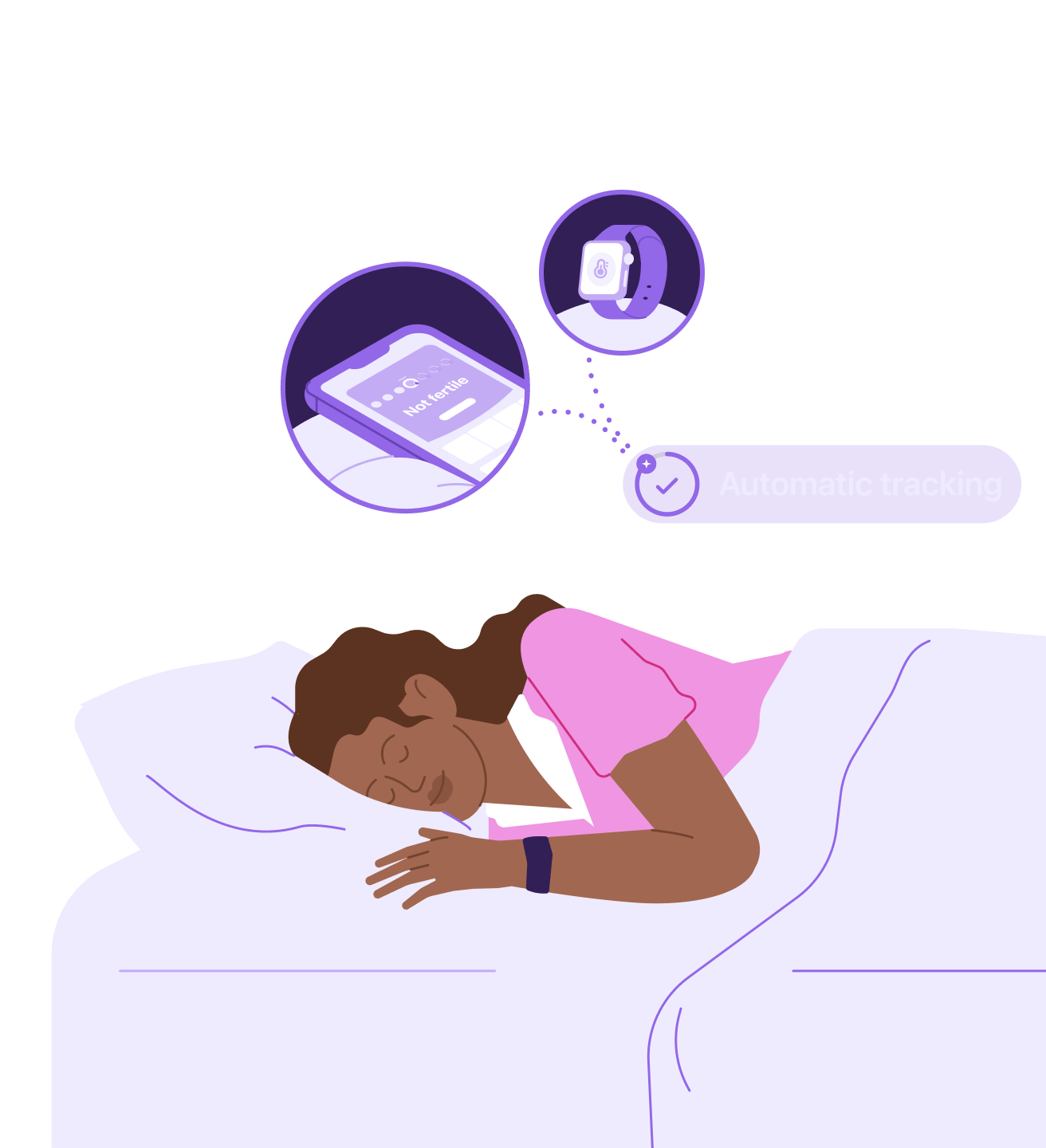
Period tracking and birth control in one place
You log your period, then your Apple Watch tracks your temperature automatically while you sleep. No travel. No pharmacy refills. Just your daily pregnancy risk, delivered.

Eligibility requirements
You could be a great fit for the study if you:
- Own an Apple Watch (Series 8, 9, 10 or Ultra)
- Don’t plan to use hormonal birth control during the study
- Are 18 years of age or older
- Are not currently pregnant or trying to conceive
Flo is working with Lindus Health to conduct this hormone-free, investigational birth control study.
Complete a short 3-minute questionnaire to see if you’re eligible.
How the study works
0-2 months
- Check eligibility and consent to data collection. Your data is safe. See our privacy policy
- Log your next period in the app. You’re now enrolled!
- Connect your Apple Watch. Wear it while you sleep.
- View your daily pregnancy risk and use protection accordingly.
2-18 months
- Keep logging periods and wearing your watch to bed
- Get paid to answer 5-minute monthly surveys
- Check your daily pregnancy risk and use protection accordingly. You can leave the study at any time.
What is a clinical study?
A clinical study is a research study that helps determine the safety and effectiveness of new medical treatments, devices, or technologies.
According to the FDA, Digital Contraception can be *93% effective for typical use at preventing pregnancy, compared with the pill at 93% and male condoms at 87%. This study will evaluate the effectiveness of Flo’s particular Digital Contraception, which uses Apple Watch temperature data and menstrual cycle tracking to predict pregnancy risk.
*Reference “Contraception and Birth Control Methods.” Centers for Disease Control and Prevention. 5 Aug, 2024. “Birth Control” FDA Office of Women’s Health, 10 May, 2024
Who is Flo?
Flo Health is a leading women's health technology company and creator of the Flo app, used by over 380 million women worldwide for period and cycle tracking. Flo Digital Contraception turns your period tracker into a digital form of birth control.
Who is Lindus?
Lindus is a research organization that manages clinical trials. Flo is partnering with Lindus Health to conduct this study safely and effectively. That includes managing enrollment, electronic consent, monthly surveys, participant support, and communications.
How long does the study last?
The study lasts 13-18 months and you can drop out at any time. It begins after you’ve completed enrollment and then logged one period.
Do I have to go anywhere?
No. This is a virtual study. Participate entirely from home using your own Apple Watch (Series 8, Series 9, Series 10, or Ultra) and the Flo app. No clinic visits required.
Do I own the right watch?
Check if you have the right watch to sign up for the study:
- Open the Watch app on your paired iPhone.
- Tap the My Watch tab
- Go to General > About
- Find the Model Number field and tap it until it shows a five-digit number starting with "A"
- Check it’s one of the following: A2770, A2771, A2772, A2774, A2978, A2981, A2980, A2997, A2999, A2622, A3335, A3337, A3281, or A2986
What is temperature-based birth control?
Temperature-based birth control tracks changes in your body temperature to identify when you're at risk of getting pregnant. After ovulation, your temperature rises slightly, signaling lower pregnancy risk. This study evaluates the effectiveness of combining temperature tracking from your Apple Watch with period logging in the Flo app to provide a hormone-free birth control option. This gives women another way to prevent pregnancy that works with their body, not against it.
What do I do on high-risk days versus low risk days?
On low pregnancy risk days, the Flo app shows you're less likely to get pregnant. On high pregnancy risk days, the app alerts you to use barrier methods (like condoms) or abstain from sex to prevent pregnancy. You'll check your daily pregnancy risk in the investigational app and make informed decisions about sex based on what you see. Note that Flo Digital Contraception does not protect against STIs.
Can I leave the study?
Yes. You can opt out of the study at any time via the Flo app or by informing Lindus Health in one of the clinical study surveys.